 
When applied to the blast furnace, it has increased the net profit in the production of pig iron from $0.50 to $0.70 per ton, and in the textile mill it has increased the output from 5 to 15 per cent, at the same time greatly improving the quality and the hygienic conditions surrounding the operative. The application of this new art to many varied industries has been demonstrated to be of greatest economic importance. K).A specialized engineering field has recently developed, technically known as air conditioning, or the artificial regulation of atmospheric moisture.S s = specific entropy of moist air at saturation, kJ/(kg da In Table 2, s d a is assigned a value of 0 at 0☌ and standard atmospheric pressure. S d a = specific entropy of dry air, kJ/(kg da K)Ĭ p,s = specific heat capacity of moist air at saturation, kJ/(kg.H s = specific enthalpy of moist air at saturation, kJ/kg da.Ĭ p,da = specific heat capacity of dry air, kJ/(kg In Table 2, h da is assigned a value of 0 at 0☌ and standard atmospheric pressure. H da = specific enthalpy of dry air, kJ/kg da. V s = specific volume of moist air at saturation, m 3/kg da. V da = specific volume of dry air, m 3/kg da. At given values of temperature and pressure, humidity ratio W can have any value from zero to W s. W s = humidity ratio at saturation, kg w/kg da gaseous phase (moist air) exists in equilibrium with condensed phase (liquid or solid) at given temperature and pressure (standard atmospheric pressure). The following properties are shown in Table 2: t = Celsius temperature,, based on the ITS-90 and expressed relative to absolute temperature T in Kelvin, by the following relation: 2009, 2020), shows values of thermodynamic properties of saturated moist air and dry air at 101.325 kPa and temperatures from −60 to 90☌. Table 2, calculated using ASHRAE’s (2021) LibHuAirProp software (based on ASHRAE RP-1485 Hermann et al. The gas constant for dry air using the Mohr and Taylor (2005) value for the universal gas constant is Using the projected changes, the relative molecular mass for dry air for at least the first half of the 21st century is 28.966 kg da/kmol, based on the carbon-12 scale. 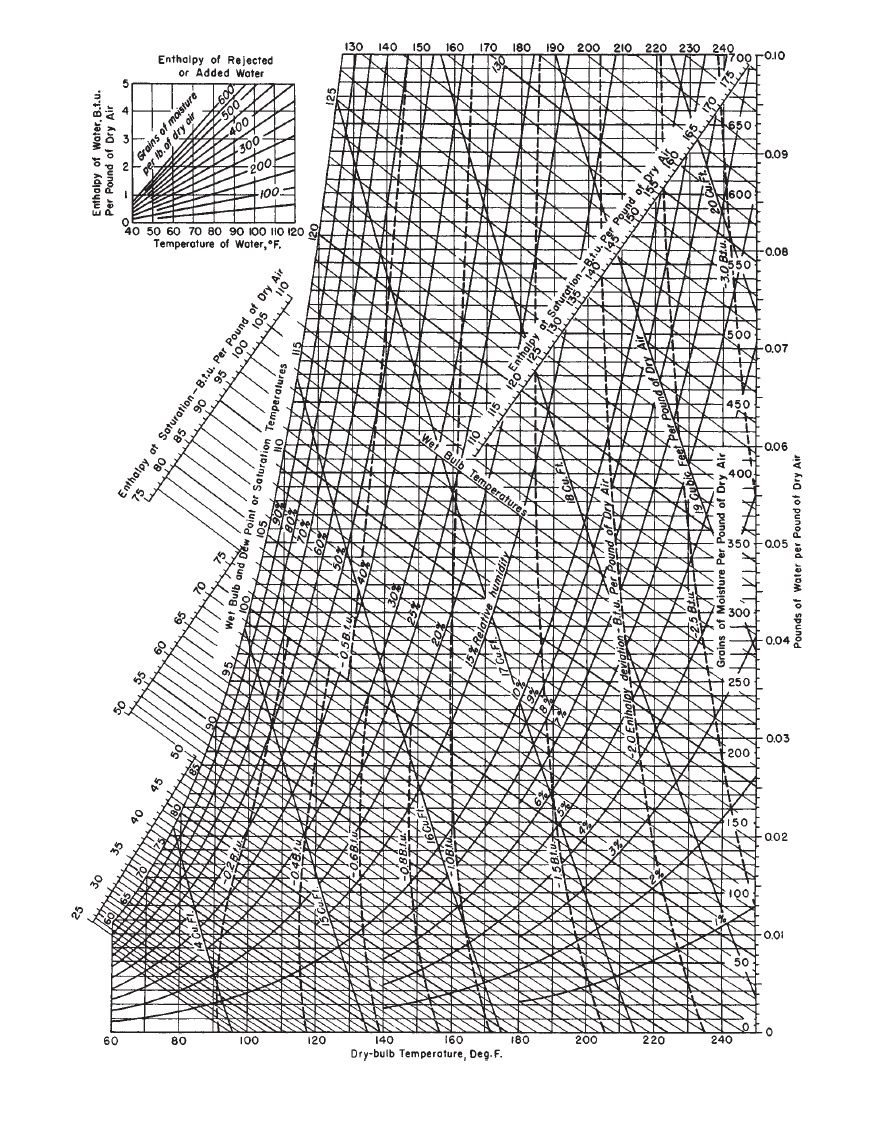
Increases in carbon dioxide are offset by decreases in oxygen consequently, the oxygen percentage in 2036 is projected to be 20.9352. Carbon dioxide reached 0.0379 in 2005, is currently increasing by 0.00019 percent per year and is projected to reach 0.0438 in 2036 (Gatley et al. Harrison (1965) and Hyland and Wexler (1983a) used a value 0.0314 (circa 1955) for carbon dioxide. Harrison (1965) lists the approximate percentage composition of dry air by volume as: nitrogen, 78.084 oxygen, 20.9476 argon, 0.934 neon, 0.001818 helium, 0.000524 methane, 0.00015 sulfur dioxide, 0 to 0.0001 hydrogen, 0.00005 and minor components such as krypton, xenon, and ozone, 0.0002. Its composition is relatively constant, but small variations in the amounts of individual components occur with time, geographic location, and altitude. Atmospheric air contains many gaseous components as well as water vapor and miscellaneous contaminants (e.g., smoke, pollen, and gaseous pollutants not normally present in free air far from pollution sources).ĭry air is atmospheric air with all water vapor and contaminants removed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
